|
Overall, the periodic table with charges is useful to help determine the nature of the chemical element in its ionic form, i.e., whether it loses or gains electrons to achieve the nearest inert gas configuration. One can use the periodic table to predict whether an atom will form an anion or a cation, and you can often predict the charge of the resulting ion. The relationship between ionic charge and the number of valence electrons for an element is an essential concept in chemistry. For example, helium (with atomic number 2) has a complete first shell, while in case of neon with atomic number 10, 2 electrons fill the first shell and the remaining 8 occupy the second shell, thus making the element inert. The Roman numeral in each name represents the charge on the ion and. In case of inert gases or noble gases (the helium group), the electrons fill up the shell completely and their charge is zero. Image of a periodic table showing the charges on the monatomic cations: +1 for. So, to achieve a complete shell (nearest inert gas, neon like configuration), it donates its 1 electron, thus having a charge +1 (represented as Na 1+). 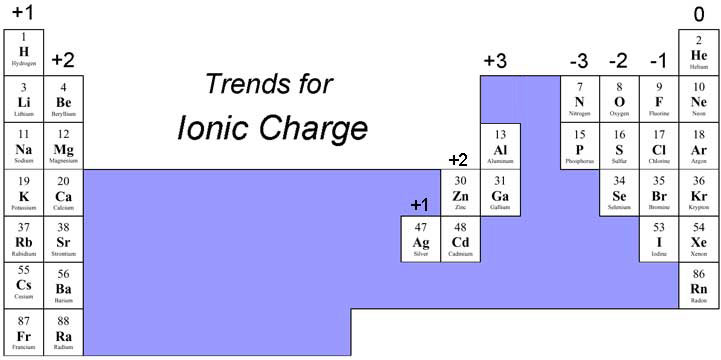
Over here, the electronic configuration is 2 electrons in the S shell, 8 in the P shell, and remaining 1 in the outer D shell. ionic charge The electrical charge of an ion, created by the gain (negative charge) or loss (positive charge) of one or more electrons from an atom or group. Let’s take an example of sodium (Na) that has atomic number 11. 
Thus, according to this method, 2 electrons are allotted in the first shell, 8 electrons in the second shell, and 18 electrons in the third shell. Positively charged ions are called cations negatively charged ions, anions. It is possible to estimate the charge that an ion will have from the position the element occupies in the periodic table of elements. 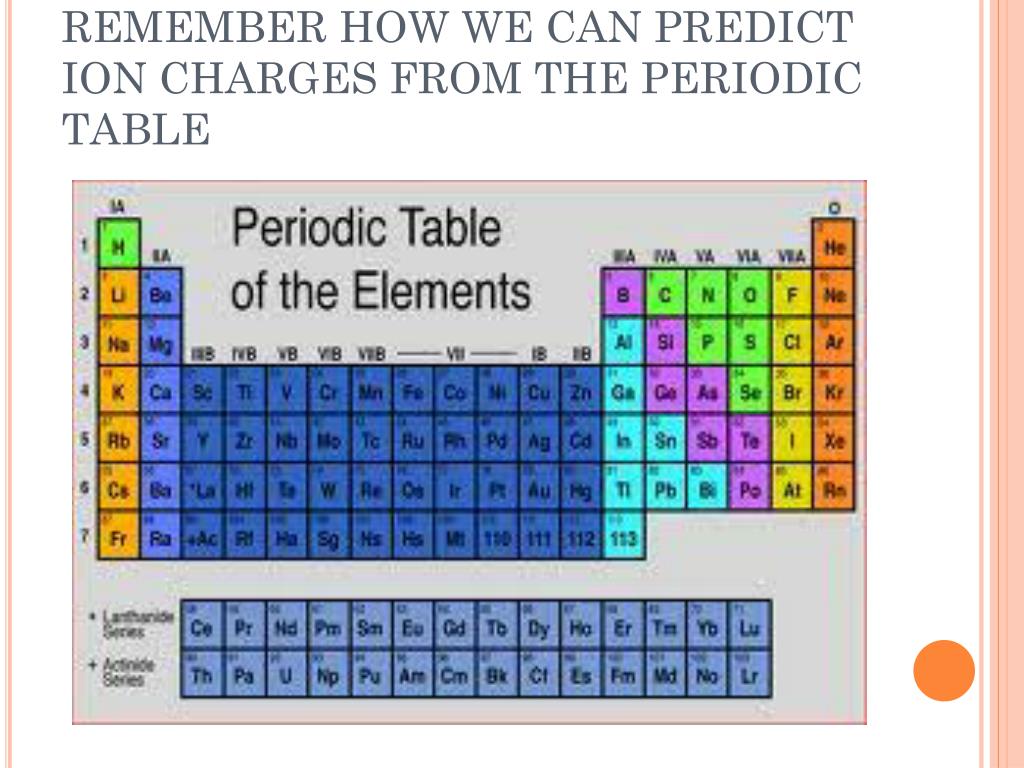
In order to find out the charge, first examine the configuration of electrons in the orbitals or shells (S, P, D, and F shells) as per the 2n 2 rule (where ‘n’ is the number of shells). ion, any atom or group of atoms that bears one or more positive or negative electrical charges.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
